
 Go to 2002 RSC Annual Report Index
Go to 2002 RSC Annual Report Index
Organic Chemistry
Organic Synthesis, Methodology and Host-Guest Chemistry
Dr Mick Sherburn
http://rsc.anu.edu.au/research/sherburn.php
Cascade (domino) reactions are spectacular events in which many bonds are made and broken in a single step. These reactions hold much promise for achieving more efficient syntheses: a pressing need in times of increasing production costs and the importance of protecting the environment by reducing waste. Our research program involves the design and implementation of sequences of cycloaddition reactions, free radical reactions and transition metal-mediated reactions to prepare polycyclic molecules with important biological properties. This program also targets new ways to achieve molecular recognition, complexation and catalysis. Overall, the primary goal is to synthesise such complex molecules in a practical manner.
Efficient Total Synthesis: Anti-tumour and Anti-Alzheimer's Natural Products
Lignans like podophyllotoxin have cancer-fighting properties and are used in chemotherapy. An efficient and highly modular approach for the synthesis of these compounds has been developed. This strategy has several advantages over previous syntheses, the most significant of which being that it allows a high level of convergency at the end of the synthetic route.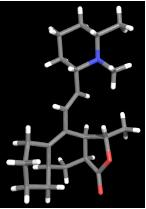 Himbacine
is natural product isolated from Galbulimima baccata, a
species of tree found in Northern Australia and Papua New Guinea.
Himbacine was found to exhibit strong, selective binding to
muscarinic receptors of the M2 subtype. Speculation that selective
presynaptic muscarinic receptor antagonists might find application in
the treatment of neurodegenerative disorders such as Alzheimer's
disease provoked our interest in Galbulimima alkaloids. A modular
strategy for the synthesis of himbacine and analogues has been
developed. The synthesis draws upon new chemistry developed in other
projects currently under way in the group. (with J. Fischer,
A.J. Scott, L.A. Sharp, L.S.-M. Wong, A.J. Reynolds
[U. Sydney])
Himbacine
is natural product isolated from Galbulimima baccata, a
species of tree found in Northern Australia and Papua New Guinea.
Himbacine was found to exhibit strong, selective binding to
muscarinic receptors of the M2 subtype. Speculation that selective
presynaptic muscarinic receptor antagonists might find application in
the treatment of neurodegenerative disorders such as Alzheimer's
disease provoked our interest in Galbulimima alkaloids. A modular
strategy for the synthesis of himbacine and analogues has been
developed. The synthesis draws upon new chemistry developed in other
projects currently under way in the group. (with J. Fischer,
A.J. Scott, L.A. Sharp, L.S.-M. Wong, A.J. Reynolds
[U. Sydney])
analogue of himbacine,
prepared for biological testing
A Deeper Understanding of the Most Important Organic Reaction
The Diels-Alder reaction is one of the most powerful and most commonly used reactions in synthetic organic chemistry. Predicting, controlling and explaining the stereochemical outcome of its intramolecular variant continues to be a major activity within the group. The location of transition structures at high levels of theory is providing stimulating new insights into the reaction. This deeper understanding is driving the development of new methodology. We have developed a novel, efficient and very general way to produce complex polycyclic molecules with useful biological properties from simple, unsaturated, acyclic precursors using a sequence of intramolecular Diels-Alder reactions. (with T.N. Cayzer, N. Miller, C.I. Turner, L.S. M. Wong [U. Sydney], M.N. Paddon-Row [U. NSW])
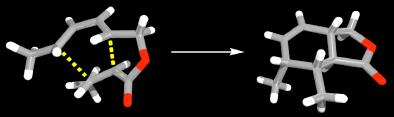
A computed transition structure along with the X-ray crystal
structure of its corresponding cycloaddition product
Host-guest Chemistry
Research in this area is concerned with the design and synthesis of host molecules based upon cavitands (rigid, bowl-shaped molecules) for molecular recognition, complexation and catalysis. Investigations into potential uses of these intriguing hosts as molecule-sized devices is under way. (with E.S. Barrett, J.R. Hansen [U. Sydney])
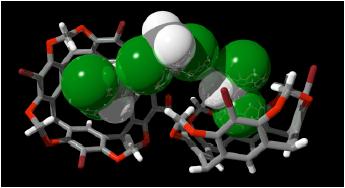
A model of one of our hosts (a bis-bowl molecule, represented in wire frame format) binding three guest molecules (dichloromethane, represented as space filling models) in the solid state.